Dr. Attaman is offering Regenexx SD Bone Marrow Concentrate (BMC) stem cell treatment for degenerative disc disease in Bellevue and Seattle.
We are the only licensed Regenexx clinic in Washington!
Review of “Percutaneous injection of autologous bone marrow concentrate cells significantly reduces lumbar discogenic pain through 12 months” from the journal Stem Cells
Degenerative disc disease (DDD) affects millions of people worldwide and has a heavy impact on both patients and healthcare providers. Damaged or inflamed tissues are generally treated through invasive surgery and drug regimens, although stem cell based treatments now represent an improved alternative strategy. Bone marrow concentrated cells (BMC) contain a variety of stem and progenitor cells, including mesenchymal stem cells (MSC) [1, 2], known to positively affect the nucleus pulposus cells of intervertebral discs [3]. Now, researchers from the University of Texas at Austin and Celling Biosciences have assessed autologous BMC stem cells for their ability to treat moderate to severe discogenic low back pain [4].
Pettine et al. first estimated the MSC content of patient-derived BMC samples to be around 0.0025% or 2,713 (± 491) per mL. Non-invasive percutaneous Injection of the autologous BMC into the symptomatic disc(s) in the 26 patients enrolled in the study led to a significant decrease in various pain scores (Pre‐treatment Oswestry Disability Index (ODI) and Visual Analogue Scale (VAS)) at 3, 6 and 12 months. Furthermore, 8 out of 20 patients displayed improvements when scored on a magnetic resonance imaging (MRI) classification system for lumbar disc degeneration. The attached figure shows representative MRI images before treatment (A) and 12 months after injection (B) of autologous BMCs into two intervertebral discs. Interestingly, patients receiving concentrations of MSCs at levels higher than 2,000 cells per mL improved significantly faster and had significantly reduced pain levels as compared to patients receiving concentrations lower than 2,000 cells per mL. The authors report no deleterious side effects associated with the initial injection, or the injected cells themselves.
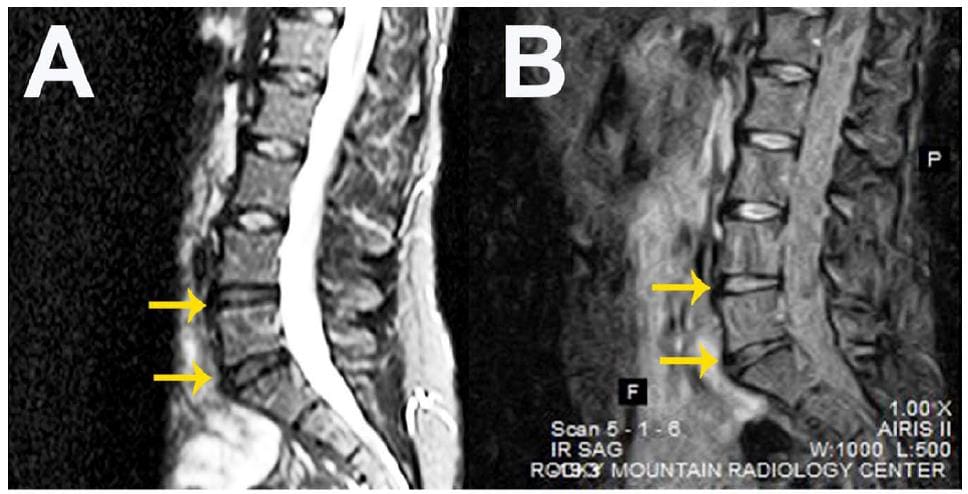 The study demonstrates that Regenexx SD BMC injection appears to be safe, feasible, and effective in the treatment of DDD, and highlights the importance of MSC concentration on this effect. Specifically, the treatment significantly reduced pain in patients with moderate to severe discogenic pain to a level comparable with artificial disc replacement (See original study for extensive references). Altogether, this signifies another great success for MSC-based regenerative medicine using a minimally-invasive technique, and is an advancement on previous cell-based treatments for lower back pain [5, 6] which will no doubt promote larger clinical trials with an expanded patient population to further assess this approach.
The study demonstrates that Regenexx SD BMC injection appears to be safe, feasible, and effective in the treatment of DDD, and highlights the importance of MSC concentration on this effect. Specifically, the treatment significantly reduced pain in patients with moderate to severe discogenic pain to a level comparable with artificial disc replacement (See original study for extensive references). Altogether, this signifies another great success for MSC-based regenerative medicine using a minimally-invasive technique, and is an advancement on previous cell-based treatments for lower back pain [5, 6] which will no doubt promote larger clinical trials with an expanded patient population to further assess this approach.
Another Study:
References
- Murphy MB, Moncivais K, and Caplan AI Mesenchymal stem cells: environmentally responsive therapeutics for regenerative medicine. Exp Mol Med 2013;45:e54.
- Murphy MB, Blashki D, Buchanan RM, et al. Adult and umbilical cord blood-derived platelet-rich plasma for mesenchymal stem cell proliferation, chemotaxis, and cryo-preservation. Biomaterials 2012;33:5308-5316.
- Sobajima S, Vadala G, Shimer A, et al. Feasibility of a stem cell therapy for intervertebral disc degeneration. Spine J 2008;8:888-896.
- Pettine KA, Murphy MB, Suzuki RK, et al. Percutaneous injection of autologous bone marrow concentrate cells significantly reduces lumbar discogenic pain through 12 months. Stem Cells 2014;
- Orozco L, Soler R, Morera C, et al. Intervertebral disc repair by autologous mesenchymal bone marrow cells: a pilot study. Transplantation 2011;92:822-828.
- Coric D, Pettine K, Sumich A, et al. Prospective study of disc repair with allogeneic chondrocytes presented at the 2012 Joint Spine Section Meeting. J Neurosurg Spine 2013;18:85-95.
